 
The reason is that they usually appear in multiple reactants and products, so if you tackle them first you're usually making extra work for yourself. When balancing chemical equations, the last step is to add coefficients to oxygen and hydrogen atoms. Iron is balanced, with 4 atoms of iron on each side of the equation. By inspection (i.e., looking at it), you know you have to discard a coefficient of 2 for some higher number.ģ Fe doesn't work on the left because you can't put a coefficient in from of Fe 2O 3 that would balance it.Ĥ Fe works, if you then add a coefficient of 2 in front of the rust (iron oxide) molecule, making it 2 Fe 2O 3. While that would balance iron, you already know you're going to have to adjust oxygen, too, because it isn't balanced. There is one atom of iron on the left and two on the right, so you might think putting 2 Fe on the left would work. Iron is present in one reactant and one product, so balance its atoms first. Balance any oxygen or hydrogen atoms last.Balance atoms present in a single molecule of reactant and product first.Basically, you look at how many atoms you have on each side of the equation and add coefficients to the molecules to balance out the number of atoms. There is a strategy that will help you balance equations more quickly. 
As with subscripts, you don't write the coefficient of "1", so if you don't see a coefficient, it means there is one molecule. If, for example, you write 2 H 2O, that means you have 2 times the number of atoms in each water molecule, which would be 4 hydrogen atoms and 2 oxygen atoms. 
When balancing equations, you never change subscripts. You add coefficients. Coefficients are whole number multipliers. Remember that if there's no coefficient in front of an element, it's assumed that the coefficient is 1.Add Coefficients To Balance Mass in a Chemical Equation Now the number of atoms in each element is the same on both sides of the equation, so the equation is balanced. To balance this, add the coefficient 2 before H2 on the left side of the equation so there are 4 hydrogen atoms on each side, like 2H2 + O2 → 2H2O. However, subscripts can't be changed and are always multiplied by the coefficient, which means there are now 4 hydrogen atoms on the right side of the equation and only 2 hydrogen atoms on the left side. For the equation H2 + O2 → H2O, you would add the coefficient 2 before H2O on the right side so that there are 2 oxygen atoms on each side of the equation, like H2 + O2 → 2H2O. 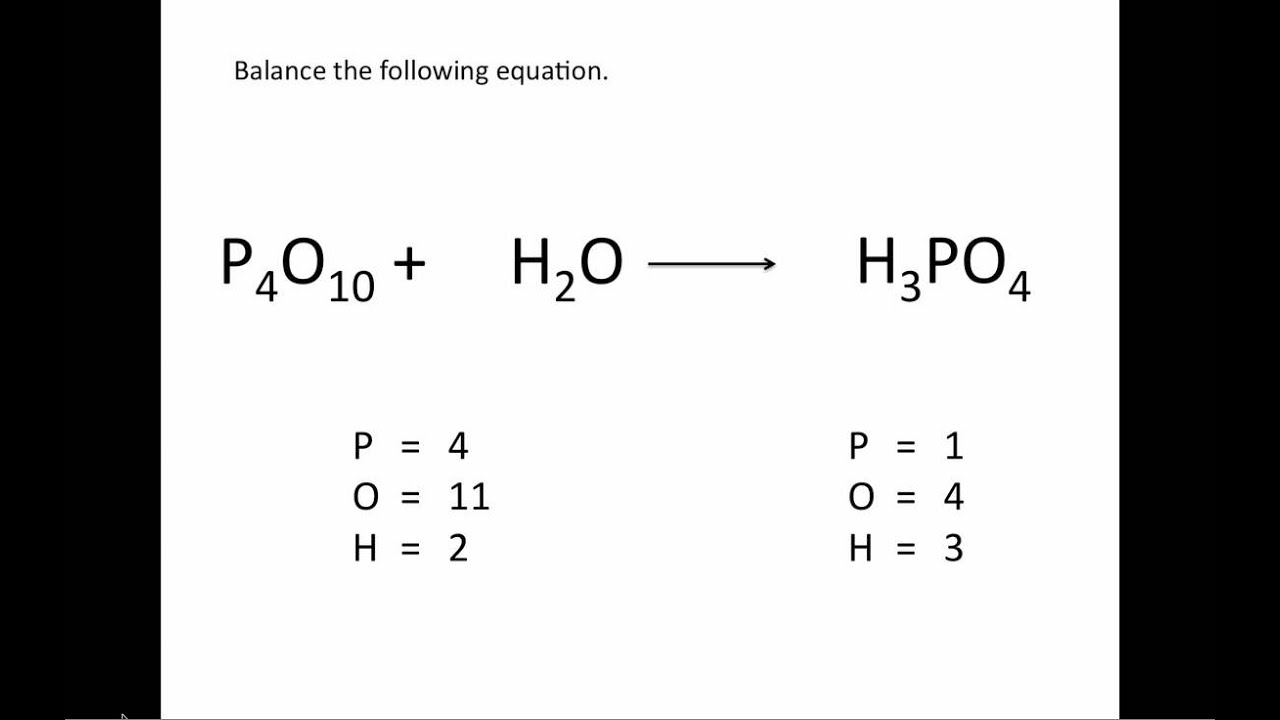
To balance the equation, you'll need to add coefficients to change the number of atoms on one side to match the other. Since the number of atoms in each element isn't identical on both sides, the equation is not balanced. There are 2 hydrogen atoms and 1 oxygen atom on the right, so you would write "H=2" and "O=1" under the right side. For the equation H2 + O2 → H2O, there are 2 hydrogen atoms being added to 2 oxygen atoms on the left, so you would write "H=2" and "O=2" under the left side. For example, your equation should look something like "H2 + O2 → H2O." Count the number of atoms in each element on each side of the equation and list them under that side. To balance a chemical equation, first write out your given formula with the reactants on the left of the arrow and the products on the right.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
